Certain inflammatory and infectious diseases, including atherosclerosis and tuberculosis, are caused by the accumulation inside immune cells of harmful substances, such as lipids and bacteria. A multidisciplinary study published in Proceedings B of the Royal Society, by researchers from the Universities of Oxford and Sydney, has shown how cell cannibalism contributes to this process.
Hugh Ford, an applied mathematics PhD student with Prof. Mary Myerscough at the University of Sydney, conducted the research whilst a visiting student at Oxford Mathematics. With Prof Myerscough and Prof. Helen Byrne in the Oxford Mathematics’s Wolfson Centre for Mathematical Biology, Hugh developed a mathematical model that accurately describes the accumulation of harmful substances in macrophages, a type of white blood cell, which act as “waste-disposal” cells for the immune system. When there are too many of these substances for macrophages to handle, they are unable to remove the excess from the area before they die. This triggers a cycle in which macrophages that die while removing harmful substances from the arteries leave the substances in situ, causing more macrophages to be recruited to ingest them. The researchers’ model describes how this leads to an accumulation of macrophages. These ingest the dead cells, along with the cholesterol they have accumulated, causing the cycle to accelerate.
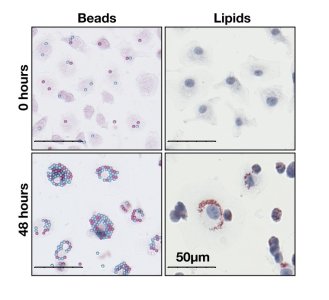
The researchers tested their model experimentally, in collaboration with Prof. David Greaves at the Dunn School of Pathology in Oxford. Hugh tracked the accumulation of plastic beads in thousands of macrophages in laboratory experiments, generating data to validate the cascading effect of substance accumulation predicted by the mathematical model. Dr Joshua Bull, an Oxford Mathematics postdoctoral researcher, assisted with the data analysis by adapting his existing image analysis software to enable automatic counting, from thousands of high-resolution images, of individual macrophages and the numbers of plastic microbeads contained within them.
Prof. Greaves from the Dunn School of Pathology said: “Hugh’s mathematical modelling allowed us to do a set of biology experiments that shed new light on the processes that drive diseases. Armed with these new insights we are keen to look for drugs that enhance tissue protection by changing cell behaviour.”
Hugh said the paper contributes to a growing body of evidence that casts cannibalistic cell removal as a double-edged sword. “While on the one hand this process is crucial for tissue stability and the resolution of inflammation, it also perpetuates subcellular accumulation of harmful substances that can then contribute to the development of diseases, such as heart disease and tuberculosis,”