New methods for localising radiation treatment of tumours depend on estimating the spatial distribution of oxygen in the tissue. Oxford Mathematicians hope to improve such estimates by predicting tumour oxygen distributions and radiotherapy response using high resolution images of real blood vessel networks.
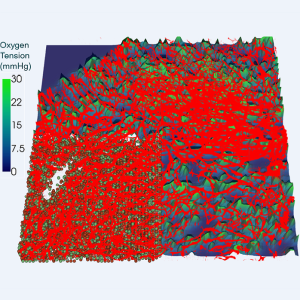
Oxygen distributions in living tissues depend on the positioning of the smallest blood vessels, the capillaries. In healthy tissue the capillary spacing is tightly regulated and vessels are evenly spaced. In diseased tissue, such as tumours, the capillary spacing can be heterogeneous, with large regions left with no vessels. In the clinic, we can estimate the capillary distribution using imaging techniques such as functional MRI, which is then used to target radiation. However, MRI can’t resolve small-scale variations in capillary spacing, potentially leaving pockets of low oxygen, radiotherapy resistant cells undetected.
Mathematical modelling can be used to study how important small-scale, undetectable, variations in capillary spacing are to overall radiotherapy response. If they are relatively unimportant it may be possible to use lower radiation doses and reduce damage to nearby tissues. Thanks to the recent development of high-resolution imaging techniques by colleagues Bostjan Markelc and Ruth Muschel in the CRUK/MRC Oxford Institute for Radiation Oncology, Oxford Mathematicians James Grogan, Anthony Connor (former), Philip Maini and Helen Byrne and Joe Pitt-Francis in the Department of Computer Science have been able to use real, 3D, vessel data to model tumour oxygen distributions and radiotherapy responses for the first time.
Their study, recently published in the Transactions on Biomedical Engineering, involved high resolution multi-photon imaging of well-vascularized murine colon adenocarcinoma (MC38) tumours in mice. The images were used to extract 3D blood vessel structures, which were used as inputs to multi-scale, multi-physics mathematical models of oxygen transport, tumour growth and radiation therapy. The models predicted that radiotherapy outcome was relatively insensitive to small-scale (order of 1 mm) variations in capillary spacing for the studied, well-vascularized tumour. They also demonstrated that previous 2D models, which used artificial tumour vessel data, overestimated the sensitivity of radiotherapy response to heterogeneities in capillary spacing. The newly predicted lack of sensitivity to variations in capillary spacing should help inform the on-going design of localized radiotherapy treatments. However, further work is required to determine whether predictions extend to other tumour types, notably in humans which can have different vessel network structures to the studied mice.
Some of the main challenges in the study were the processing of 3D imaging datasets for integration with the mathematical models and the development of software for multi-scale, multi-physics simulation of tumour growth in large 3D domains. The resulting software is based on the open-source Chaste library, developed in the Mathematical Institute and Department of Computer Science, University of Oxford. The authors are now applying their integrated imaging and modelling approach to several related problems in the area of vascularized tumour treatment.
For further demos see the software page.